Barrovian Facies
Steven Dutch, Professor Emeritus, Natural and Applied Sciences, Universityof Wisconsin - Green Bay
The Barrovian facies series, named for George Barrow, who described them around 1900,is the "normal" metamorphic facies series. These are the rocks formed in atypical geothermal gradient of about 25 C per km. The typical pressures under which theserocks form are 5-10 kb (mid-crustal depths). All temperatures are approximate and dependent on pressure.
Greenschist Facies
400 C - Quartz-Albite-Muscovite-Chlorite Subfacies
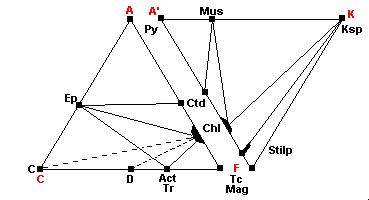 | The phyllosilicate stilpnomelane is present: Stilp = K1-x(Fe",Mg,Al)3-xSi4O10(OH)2.nH2O Carbonate-rich rocks may contain dolomite or calcite along with chlorite (dashed lines). Magnesite may be present in magnesium rich rocks. |
450 C - Quartz-Albite-Epidote-Biotite Subfacies
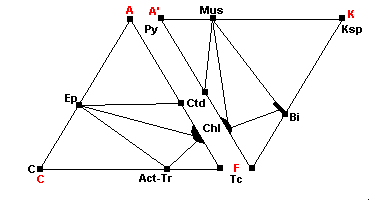 | Stilpnomelane breaks down: Stilp + musc = Bi + Chl + Qz K-feldspar no longer coexists with chlorite: Chlorite and calcite are no longer compatible: |
500 C - Quartz-Albite-Muscovite-Almandine Subfacies
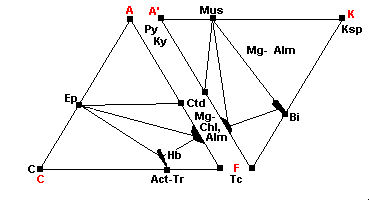 | Chlorite begins to break down, releasing iron to form almandine: (Fe,Mg)-Chl + Qz = Alm + Mg-Chl It also releases iron to form hornblende: |
Almandine-Amphibolite Facies
550 C - Staurolite-Almandine Subfacies
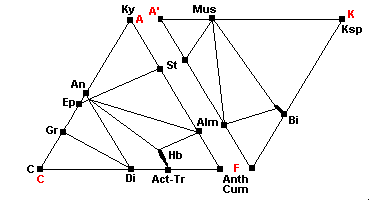 | The breakdown of chlorite is complete, chloritoid disappears and staurolite appears: Fe-Chl + Musc = St + Bi + Alm + H2O Ctd + Chl + Mus = St + Bi + Alm + H2O If there are appreciable volatiles, scapolite may form in addition to plagioclase. The scapolites are a solid solution series of marialite (3albite + NaCl) and meionite (3anorthite + CaCO3 or CaSO4. |
600 C - Kyanite-Almandine-Muscovite Subfacies
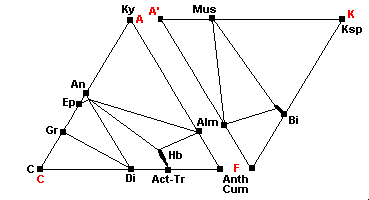 | Staurolite is no longer stable and disappears: 3St + 2Qz = Alm + 5Ky + H2O St + Musc + (Mg,Fe)-Bi = Ky/Sil + Fe-Bi + H2O (Staurolite breaks down and the reaction yields more iron-rich biotite) |
680 C - Sillimanite-Almandine-Orthoclase Subfacies
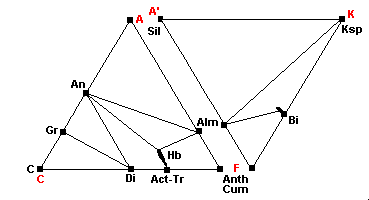 | Kyanite transforms to sillimanite Microcline transforms to orthoclase Muscovite becomes unstable: Epidote is no longer stable: |
Return to Mineralogy-Petrology Index
Return to Thin-Section Index
Return to Crystals and Light Index
Return to Crystal Structures Index
Return to Mineral Identification Tables
Return to Professor Dutch's Home Page
Created 22 Sept 1997, Last Update